This application note was produced in collaboration with the Leake Lab at the University of York.
In this application note, we explore work by Howard et al., who used single-molecule Förster Resonance Energy Transfer (smFRET) and fluorescence correlation spectroscopy (FCS) on the EI-FLEX to reveal two previously unknown, salt-influenced conformational states on the bacterial helicase Rep1. X-ray crystallography had identified two conformational states, open and closed structures, respectively. Here, both techniques were performed on the EI-FLEX instrument, providing single-molecule insights that were complementary to several other biophysical techniques used in this work.
Overview of this application note:
- FCS identifies variations in hydrodynamic radius and diffusion time that are likely caused by salt-induced protein conformational changes
- smFRET uncovers two conformational states that were not identified in crystal structures, which are influenced by salt concentration and DNA binding
- Combining smFRET data with complementary techniques, such as ABEL trapping, resolved multiple interconversion conformational states that persisted on sub-millisecond to second timescales
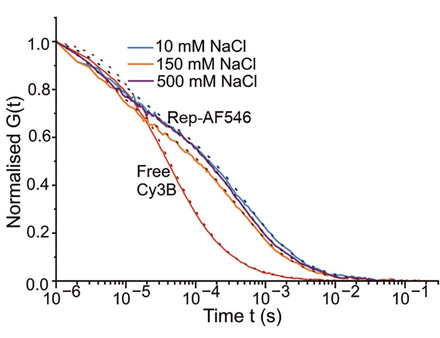
Figure 1 – Influence of NaCl on Rep helicase diffusion time
Normalised autocorrelation plotted against time for free Cy3b (control) and three concentrations of NaCl (10, 150 and 500 mM) shows diffusion time is influenced by salinity, prompting conformational analysis by smFRET.



