Ternary complexes
Directly quantify ternary complex formation with rapid single-molecule screening
Generate binding curves for ternary complexes with confidence
Characterising the formation of ternary complexes, where three distinct molecular components interact, is essential for drug discovery and systems biology. However, understanding how a third partner, such as a co-receptor, a small-molecule recruiter, or a bridging ligand, stabilises or modulates a complex presents a significant analytical challenge.
Solution-based single-molecule fluorescence assays are an ideal way to study ternary complex formation while avoiding artefacts associated with immobilised assays. The ability to visualise multiple binding partners with fluorescent labels enables direct observation of ternary complex formation and quantification of binding and dissociation events. Meanwhile, single-molecule resolution enables detection of complex heterogeneity and intermediates that may be obscured in ensemble assays.
Fluorescence Cross-Correlation Spectroscopy (FCCS), Fluorescence correlation spectroscopy (FCS) and single-molecule Förster resonance energy transfer (smFRET) provide a robust toolkit for dissecting these multi-component assemblies in their native state. FCCS is the gold standard for ternary systems. By labelling components with different fluorophores, FCCS detects the synchronised movement of molecules, providing a direct measurement of ternary complex concentration, while single-colour FCS provides kinetic insights of binary components. Additionally, smFRET can detect the precise inter-molecular distances and conformational shifts that occur when a binary pair transitions into a ternary state.
Characterise ternary complexes in physiologically relevant environments
- Directly quantify the ternary fraction, distinguishing full complexes from binary intermediates and unbound monomers
- Characterise a variety of ternary complexes, from PROTACs and molecular glues, to bispecific antibodies and DNA origami systems
- Rapidly detect the hook effect, identifying the optimal dose where ternary complex formation is maximised before autoinhibition occurs
- Measure cooperativity, revealing whether the binding of a primary ligand induces a favourable conformational change in the target protein
- Determine individual dissociation constants for each interface within the complex
- Identify transient intermediates during ternary complex formation, enabling differentiation between productive and unproductive pathways and complexes
- Study ternary complex stability and kinetics in solution, using physiologically relevant matrices such as cell lysates and serum
Cross-correlation provides a direct readout of ternary complex formation
This figure illustrates the ability of FCCS to directly measure ternary complex formation with three DNA oligos. When both fluorescently labelled oligos are bound to oligo 2, they co-diffuse through the confocal volume, resulting in higher cross-correlation as the concentration of oligo 2 is increased. The increase in cross-correlation is proportional to the increase in the concentration of oligo 2 until the hook effect is reached at around 15 nM of oligo 2.
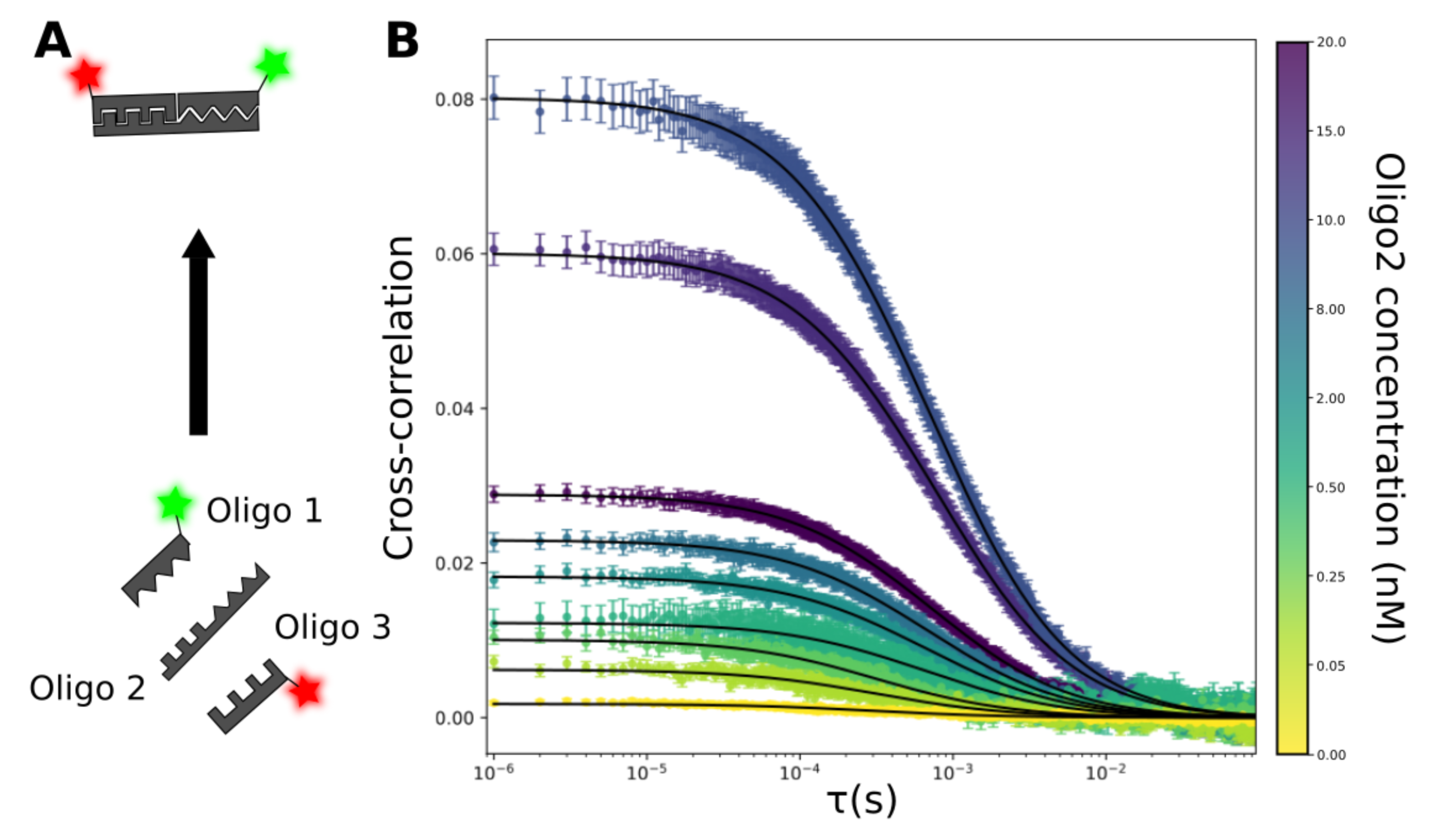
(A) Schematic of ternary complex formation of oligo 1 (green label), oligo 2 (no label) and oligo 3 (red label)
(B) Cross-correlation measured on the EI-FLEX with 20nM oligos 1 and 3 and increasing concentrations of oligo 2 (coloured datapoints). All measurements were acquired in TE buffer supplemented with 150mM NaCl and 1% glycerol. Data was acquired and analysed in-house at Exciting Instruments.
Ternary complex papers and
pre-prints featuring EI-FLEX data
Protein structure and dynamics FAQs
Can I distinguish between a binary and a ternary complex in a single sample?
FCCS directly identifies ternary complexes by quantifying co-diffusing molecules. Here, the co-diffusing ternary complex can be distinguished from the singly labelled binary intermediates or monomer species. If the third, unlabelled subunit is not obligatory for two labelled species to interact, codiffusion should also be quantified in the absence of the unlabelled subunit.
Can cooperative binding be quantified?
Yes, FCCS and FCS can be used to measure cooperative binding. Additionally, FRET can be used to detect conformational changes upon binding, providing insight into the mechanism of cooperativity.
Can these techniques detect the hook effect?
Absolutely – at high concentrations of a recruiter molecule, ternary complex formation often drops off (the hook effect). FCCS is ideal for mapping this bell-shaped curve because it measures the actual concentration of the ternary species directly in solution, determining the optimal conditions for ternary complex formation, such as for PROTACS or molecular glues.
Is it possible to measure the stability of a complex in human serum?
Yes. These techniques are highly robust in physiologically relevant matrices and have proven performance in serum and diluted cell lysates.
How do I determine which part of the complex is compromising its stability?
FCCS can be used to quantify a variety of kinetic rates, including dissociation constants for individual subunits of a ternary complex.
Do these methods require immobilisation on a surface?
No. All three techniques are solution-based using our instrumentation. This is a major advantage for ternary complexes, as surface immobilisation has the potential to sterically hinder binding of one or more partners, or modulate avidity in undesired ways.
Can I detect if a third molecule is an inhibitor?
Yes. In a competitive FCCS setup, you can monitor the disappearance of the cross-correlation signal between two components as you add a third, unlabelled competitor. This permits the calculation of the IC50 of an inhibitor in a complex, multi-component environment.
Can I study complexes involving intrinsically disordered proteins (IDPs)?
IDPs often only take a defined shape when they form a ternary complex. smFRET and FCS are ideal techniques for monitoring an ordered-to-disordered transition in real-time as the third partner joins the assembly.
"What sets the EI-FLEX apart is its versatility - it's our go-to for probing kinetics and conformational changes that the other systems can't resolve. This opens up a range of research for us that was previously impossible."
Professor Edwin Antony, St. Louis University





